I’m trying to raise money for the The Leukemia & Lymphoma Society, and I promised to do a few things if we reached certain goals. I said I’d write a post explaining what tumor suppressor genes are, while wearing a pirate hat and nothing else, if we raised $5000. Shiver me timbers, I did! And it’s cold!
If you want more, go to my Light the Night fundraising page and throw money at it. I’ll write about microRNAs and cancer when we hit $7500. Note that we’re also getting matching funds from the Todd Stiefel Foundation, so join in, it’s a good deal.
There’s a basic principle in biological homeostasis (and it’s also true in cybernetics) that for every process that turns something on, you have an anti-process that turns it off. When you look at what’s going on inside the cell, you’ll often get the impression that it’s spinning its wheels — every protein, for instance, is being degraded at about the same rate that it’s being synthesized. The whole cell is in a state of dynamic equilibrium, in which it might look like the concentration of some protein is constant overall, but when you look closely, individual protein molecules are being constantly made, instantly targeted for destruction, and dismantled…only to be nearly instantly replaced by a duplicate. Everything is in a state of flux, and it looks terribly wasteful, but it means that everything is fluid and flexible and the cell is incredibly responsive to subtle cues.
Last time I wrote about oncogenes, genes that are activators of crucial cellular processes like cell division, and I told you that these play a role in cancer by sending faulty signals that switch on uncontrolled cell division. Given the above principle, you might expect that if there are gene products that turn on cell division, there ought to also be gene products that turn off cell division, a kind of anti-oncogene. And there are! They’re called tumor suppressor genes.
While acting in opposition to oncogenes, tumor suppressor genes typically exhibit another difference in behavior that sets them apart. Cancer causing mutations in oncogenes are usually dominant: that is, the mutation doesn’t just knock out the gene, it has to make a hyperactive gene, and just one mutant copy gone rogue is enough to start switching on cellular activity. Tumor suppressor gene mutations tend to be recessive.
You’ve usually got two copies of every gene. A tumor suppressor works to shut down renegade activity, and a cancer-causing mutation in one is most often simply going to be a mutation that destroys the gene — but since you’ve got two copies, it has a backup. Cancer needs to kill both copies of the tumor suppressor to escape from its anti-tumor actions.
The best known tumor suppressor gene is BRCA1, and it exhibits this behavior. BRCA1 is a good gene — it’s working to protect you from breast cancer. For most of us, every cell in our body has two functional copies of BRCA1. It’s like having two cops patrolling the beat, prepared to fight off any cancer threat, and for cancer to succeed, it has to kill off both. Even one left functional can suppress any effort by the cell to go hyperactive and proliferate. A mutation that knocks out one copy is rare, but two mutations that knock out both are even more rare — it follows two-hit kinetics, which means you multiply the probability of the two events together.
You will occasionally hear about someone being at high risk of cancer because they carry a bad allele of BRCA1 — Angeline Jolie was in the news about this recently. What has happened here is that the person was born inheriting one broken copy of the BRCA1 gene — their cells only have one cop on the beat. They aren’t born with cancer — that one copy of BRCA1 is sufficient to keep them safe — but now they are at much higher risk of an accidental mutation taking out their sole protector than if they had two.
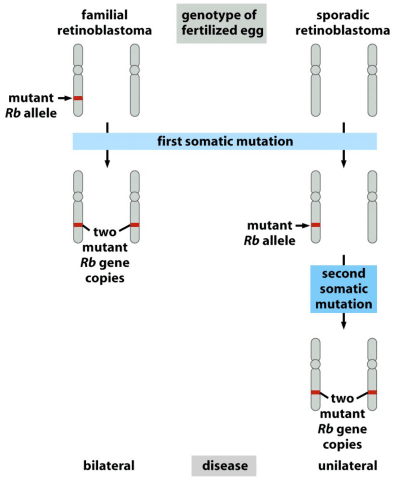
Another well known tumor suppressor gene is Rb, the gene that when mutant can lead to retinoblastoma, or cancers of the eye. It also exhibits two hit kinetics, in that familial retinoblastoma is caused by inheritance of one mutant Rb allele, so that all it takes is one mutation later in life to lead to cancer. Sporadic retinoblastoma, that is retinoblastoma without previous examples in the family, is much rarer, because it requires a first somatic mutation and a second mutation later to take out the second copy.
Having an enabling mutation in one copy at birth so greatly increases the chance of retinoblastoma that afflicted individuals are likely to have bilateral cancers affecting both eyes, while it’s so rare in sporadic cases that it is almost always unilateral, affecting only one eye.
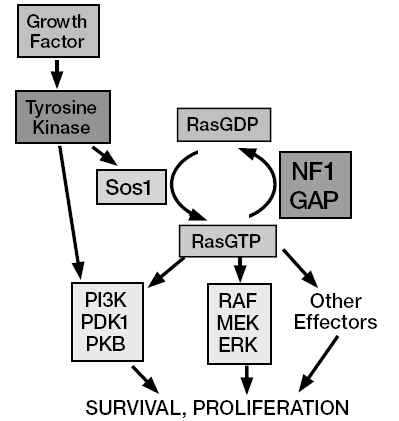
OK, you understand the principle. Last time, I wrote about the oncogene Ras, which, to make it simple switches on the mitotic machinery and promotes cell division. Does it have an anti-gene product that opposes Ras and switches off cell division?
Of course there is. Several actually. One example: recall that I told you that Ras is activated by binding GTP, and deactivated by converting GTP to GDP, and that Ras itself has GTPase activity and therefore works to switch itself off. There are also tumor suppressor proteins that are called GAPs, or GTPase activating proteins, that enhance Ras’s GTPase activity. One of them, called NF1 or Neurofibromin, binds to Ras and elevates its potency as a GTPase approximately 1000-fold — it basically makes Ras ultra-good at shutting down and doing nothing.
Oncogenes and tumor suppressors are the yin and yang of cellular regulation. Cancer cells often contain hyperactive oncogenes, and have shut down tumor suppressors…which suggests that one strategy for treating cancer is to develop inhibitors of oncogenes, and to repair, replace, or elevate expression of tumor suppressors. And that’s a rather pat explanation of some viable general strategies that of course encounter all kinds of additional complexities.
Donovan S, Shannon KM, Bollag G (2002) GTPase activating proteins: critical regulators of intracellular signaling.. Biochim Biophys Acta 1602(1):23-45.
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646-74.
Weinberg, RA (2014) Biology of Cancer. Garland Science, New York.



I’m about to terrify you.
For the pirate photo, I went looking for a big jug of grog to wave around, but we were fresh out. So I substituted another bottle from the kitchen, and cunningly concealed the label.
So I’m not wearing anything but a pirate hat, and waving…
a big bottle of olive oil.
There. That should inspire a few nightmares.
As soon as I get the bleaching done, I’ll actually read the post.
Thanks, PZ, another excellent post. Which brave soul took the photo?
I am the only soul brave enough (or inured enough) to do that. It’s a selfie, obviously.
No eyes were sacrificed in the construction of this post.
Extra virgin?
These posts have been great. Can we have more. please, even after the funds have been raised? I think all your posts are worthwhile, but I love these science explained type posts. And it makes great ammo against people who claim that eating oranges or popping vitamin X will prevent or cure cancer.
I now know more about tumor suppressor genes than I did 20 minutes ago. Thanks. Now let’s see if (given my recent medical history) I can get mine off their asses and to work doing some actual suppression.
I’ve got a question: do tumour supressors only suppress tumours, or do they have other roles as well?
I had to learn those signaling pathways in undergrad. A bit like magic formulas: Ras RAF MEK ERK…
…You’re also wearing an eyepatch and a knife in that photo.
As the post says:
They turn off cell division in general, for instance when a wound has healed or something just stops growing.
I often hear peeps say every cell in the body is replaced every five years, meaning you’re an all-new person and shit! With this article, it seems possible that’s true on the level of genes, but it still feels unlikely to me that it’s totally accurate. What’s the haps, actual experts?
…Anáil nathrach, ortha bháis is beatha, do chéal déanaimh
OK, someone get that dragon out of here before it burns the place down!
Not enough nakedness, not nearly enough nakedness.
I do appreciate the effort, however.
In the last picture the eye patch was over the left eye and in this picture the eye patch is over the right eye. In the next picture, will there be patches over both eyes?
Who says there will be a next picture? We need $2500 more.
Rich Woods at #12
Excalibur!* Right? I didn’t even look it up. Points for me if I got it right ’cause I NEVER remember stuff like this.
* Merlin’s spell for bringing the dragon, I think. Used by Morgana for evil doings.
@11
Most cells in the body are replaced within 5-7 years. Skin cells are replaced much more quickly. Bone cells last much longer, IIRC, more like 15-20 years. I think I remember that neurons do not get replaced, but I could be wrong. This is, however, from memory, when I was studying stable carbon isotopes and what they can tell us about prehistoric diets, etc. and was a decade or two ago, so I could be mistaken.
Gack! Had to try to find the e-post-it program to cover up the picture.
Still, nice clear and lucid explanation of the role of suppressor genes.
The Mythbusters showed that the eye-patch was useful if put one 20 minutes or so before boarding, and one went from bright deck to the dark below deck, changing the position of the eye-patch in the process.
Hmm…Checked in those writers who wrote in the nude. You have some famous people you emulate. Sell like #7….
HDAC inhibitors are all the rage these days for turning on oncogene expression. Unfortunately, it doesn’t work for all oncogenes. Case in point – HDAC inhibitors interfered with BRCA1 expression interrupting DNA repair.
Quite interestingly, the retinoblastoma gene plays a much larger role than “just” tumor supressor: It is the ‘restriction point’ gene which constrols the switch from G1 to S phase. It holds up replication until a certain set of requirements are satisfied such as: 1. enough glucose in the cell 2. enough amino acids 3. presence of growth factor, and presumably other things I don’t know much about such as checks for DNA damage.
Even more fascinating, Rb is disabled by HPV (the cervical-cancer causing virus) and other viruses. HPV “wants” to make immortal cells which replicate non-stop in order to produce more of the virus, and it does this by disabling the restriction point gene and causing uncontrolled replication. As a side effect, this unsurprisingly tends to cause cancer! HPV also activates telomerase, which replaces the telomeres at the ends of chromosomes and which normally function as another failsafe against uncontrolled replication.
I am unsure why supressor mutations are associated with particular locations. If we use the BRCA1 mutation as an example, doesn’t every cell have this mutated supressor? Thus, isn’t every cell that much closer to becoming cancerous?
This question brought to you by my single year of undergraduate biology, before transferring to good, clean physics.
I love these posts, thank you! If the next milestone is passed, would you use that photo as your profile picture for a year?
Seeing that picture brings to mind the need for a web filter to eliminate the risk of searing the unsuspecting cornea. it could also eliminate the danger of seeing photos of Lindsay Lohan.
How do they calculate the risk of developing certain type of cancer?
In the Angelina Jolie case, the “risk” of developing breast cancer was about 90%, right? So what does that mean? Is it an overall probability or a figure from a continuous hazard (As in a Cox model, i.e. probability of developing cancer in the next 5 years)?
I’ve read it both ways, and I wondered whether you knew.
Every cell would have a mutant copy of the tumor suppressor gene (BRCA1 in this case), but cells in different organs and tissues also have different patterns of normal gene expression too, and that includes normal differences in the pattern of expression other tumor suppressor genes, of which there are many.
Some tissues may be more vulnerable to neoplastic transformation in the setting of a germline BRCA1 mutation because they are normally more dependent on BRCA1 activity for cell cycle regulation due to their unique constitutive pattern of gene expression. Other tissues may be less vulnerable because of the expression of other tumor suppressors. Others might be totally unaffected because normally BRCA1 isn’t even expressed in those cells at all.
And, typically, most inherited mutations of tumor suppressor genes are in fact associated with a spectrum of tumors in different tissues. In Neurofibromatosis Type I, where the NF1 (neurofibromin) gene is mutated, affected individuals can get tumors in the skin, various endocrine organs, and in peripheral nerves, among others.
AFAIK you’re largely right.
Cell cycle: G1, S, G2, M. G1 is the diploid steady-state phase, G2 the tetraploid one; S is DNA synthesis, M is mitosis (normal cell division). Cells that don’t divide are in G1, which is then called G0.