The coelacanth genome has been sequenced, which is good news all around…except that I found a few of the comments in the article announcing it disconcerting. They keep calling it a “living fossil” — and you know what I think of that term — and they keep referring to it as evolving slowly
The slowly evolving coelacanth
The morphological resemblance of the modern coelacanth to its fossil ancestors has resulted in it being nicknamed ‘the living fossil’. This invites the question of whether the genome of the coelacanth is as slowly evolving as its outward appearance suggests. Earlier work showed that a few gene families, such as Hox and protocadherins, have comparatively slower protein-coding evolution in coelacanth than in other vertebrate lineages.
Honestly, that’s just weird. How can you say its outward appearance suggests it is slowly evolving? The two modern species are remnants of a diverse group — it looks different than forms found in the fossil record.
And then for a real WTF? moment, there’s this from Nature’s News section.
It is impossible to say for sure, but the slow rate of coelacanth evolution could be due to a lack of natural-selection pressure, Lindblad-Toh says. Modern coelacanths, like their ancestors, “live far down in the ocean, where life is pretty stable”, she says. “We can hypothesize that there has been very little reason to change.” And it is possible that the slow genetic change explains why the fish show such a striking resemblance to their fossilized ancestors.
Snorble-garble-ptang-ptang-CLUNK. Reset. Does not compute. Must recalibrate brain.
None of that makes sense. The modern fish do not show a “striking resemblance” to their fossilize ancestors — they retain skeletal elements that link them to a clade thought to be extinct. This assumption that Actinistian infraclass has been unchanging undermines their conclusions — the modern species are different enough that they’ve been placed in a unique genus not shared with any fossil form.
Then the argument that they must live in a stable environment with a lack of natural-selection pressure is absurd. Selection is generally a conservative process: removing selection pressures from a population should lead to an increase in the accumulation of variability. Do they mean there has been increased selection in a very narrowly delimited but stable environment?
But even that makes no sense. We should still be seeing the accumulation of neutral alleles. Increased selection is only going to remove variability in functional elements, and most of the genome isn’t. I suppose one alternative to explain slow molecular evolution would be extremely high fidelity replication, but even that would require specific selection constraints to evolve.
This article broke my poor brain. I couldn’t see how any of this could work — it ignored the fossil evidence and also seemed to be in defiance of evolutionary theory. It left me so confused.
Fortunately, though, the journal BioEssays came to my rescue with an excellent review of this and past efforts to shoehorn coelacanths into the “living fossil” fantasy, and that also explained the molecular data. And it does it plainly and clearly! It’s titled, “Why coelacanths are not ‘living fossils’”, and you can’t get much plainer and clearer than that.
First, let’s dismiss that myth of the unchanging Actinistian. Here’s a phylogeny of the coelacanth-like fossils and their one surviving species.
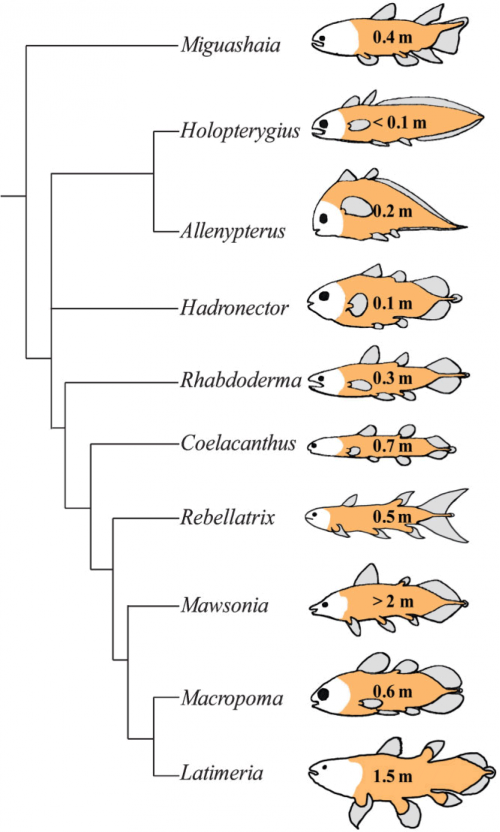
Comparison of extant and selected extinct actinistians, commonly known as coelacanths. A phylogeny of Actinistia; schematic sketches of body outlines and approximate body length (given in metre) illustrate the morphological diversity of extinct coelacanths: some had a short, round body (Hadronector), some had a long, slender body (Rebellatrix), some were eel-like (Holopterygius) whereas others resembled trout (Rhabdoderma), or even piranha (Allenypterus). Note that the body shape of Latimeria chalumnae differs significantly from that of its closest relative, Macropoma lewesiensis.
Love it. I’ve been looking for a diagram like this for a long time; creationists often trot out this claim that coelacanths haven’t changed in hundreds of millions of years, and there you can see — divergence and variation and evolution, for hundreds of millions of years.
In addition, we can look in more detail at the skull and limbs of these animals. This drawing is comparing modern Latimeria with its closest fossil relative, and even here there are dramatic differences in structure.

Comparison of the skeleton of extant and selected extinct coelacanths. A–D: Latimeria and its sister group Macropoma show numerous skeletal differences. A, B: Overall view of the skeletal organisation of the extant coelacanth and of its closest relative. A: Latimeria chalumnae. B: Macropoma lewesiensis. Relative to the body length, in L. chalumnae the vertebrae are smaller, the truncal region of the vertebral column is longer and the post anal region is shorter than in M. lewesiensis. In the latter region, the hemal arches (ventral spines) extend more ventrally in M. lewesiensis than in L. chalumnae. In addition, the swim bladder is ossified in Macropoma but not in Latimeria, and the basal bone of the first dorsal fin is characteristic of each genus. C, D: Comparison of the skulls of L. chalumnae and M. lewesiensis. C: In L. chalumnae, the mouth opens upward, the articular bone (yellow) is long and narrow, the parietonasal shield (red) is short, the premaxillary bone (orange) is devoid of denticle ornamentation, the dorsal part of the cleithum (light brown) is spiny, and the scapulocoracoid (green) is located on the ventral side. D: In contrast, in M. Lewesiensis, the mouth opens forward, the angular bone (yellow) is triangular, the parieto-nasal shield (red) is long, the premaxillary (orange) protrudes and forms a hemispherical snout which is ornamented with prominent denticles, the dorsal part of the cleithrum (light brown) is thick, and the scapulocoracoid, (green) is located more medially. Modified from [3]. E: Pectoral fin skeleton of L. chalumnae (above) and Shoshonia arctopteryx (below). The three first preaxial radials are numbered from proximal to distal. In L. chalumnae the fin appears nearly symmetrical because radial bones (orange) are arranged nearly symmetrically about the fin axis. The proximal preaxial radials 1-2 are extremely short and bear no fin ray, and the preaxial radial 3 is short and fractionated. In contrast, in S. arctopteryx the fin is strongly asymmetrical chiefly because proximal preaxial radials are long and all bear fin rays.
The authors make it clear that this idea of morphological conservation of the Actinistians is simply bogus.
In addition, an examination of the skeleton of the fossil genus Macropoma (approximately 70 Ma), the sister group of Latimeria and the only known fossil actinistian record from the Cretaceous to the present, shows some interesting differences. Not only are the extant coelacanths three times larger than their closest extinct relatives (about one and a half metres vs. half a metre), but there are also numerous structural differences. The swim bladder is ossified in Macropoma but filled with oil in Latimeria, indicating they were probably found in different types of environments. There are also noticeable differences in the vertebral column (the post anal region is shorter and ventral spines extend less ventrally in M. Lewesiensis compared with L. chalumnae), and in the attachment bones of the fins. In addition, Macropoma and Latimeria have distinctly dissimilar skull anatomies, resulting in noticeable differences in head morphology.
Finally, it should not be forgotten that external morphological resemblances can be based on a very different internal anatomical organisation. The most often emphasised resemblance between coelacanths is that they all have four fleshy-lobed-fins. Until recently, the anatomy of the lobed fins of coelacanths was only known in Latimeria, in which the pectoral fin endoskeleton is short and symmetrical. In 2007, Friedman et al. described the endoskeleton of the pectoral fin of Shoshonia arctopteryx, a coelacanth species from the mid Devonian, and therefore contemporary with Miguashaia. They showed that this earliest known coelacanth fin endoskeleton is highly asymmetrical, a characteristic that is probably ancestral since it resembles the condition found in early sarcopterygians such as Eusthenopteron, Rhizodopsis or Gogonasus. This result is additional support, if needed, that extant coelacanths have not remained morphologically static since the Devonian.
Well, so, you may be wondering, what about the molecular/genomic data? Doesn’t that clearly show that they’ve had a reduced substitution rate? No, it turns out that that isn’t the case. Some genes seem to be more conserved, but others show an expected amount of variation.
However, a closer look at the data challenges this interpretation [of slow evolution]: depending on the analysed sequence, the coelacanth branch is not systematically shorter than the branches leading to other species. In addition, most phylogenetic analyses – including analysis of Hox sequences – do not support the hypothesis that the Latimeria genome is slow evolving, i.e. they do not place coelacanth sequences on short branches nor do they detect low substitution rates. The clearest example, which involves the largest number of genes, is a phylogeny based study of forty-four nuclear genes that does not show a dramatic decrease, if any, in the rate of molecular evolution in the coelacanth lineage. What we know about the biology of coelacanths does not suggest any obvious reason why the coelacanth genome should be evolving particularly slowly.
So why is this claim persisting in the literature? The authors of the BioEssays article made an interesting, and troubling analysis: it depends on the authors’ theoretical priors. They examined 12 relevant papers on coelacanth genes published since 2010, and discovered a correlation: if the paper uncritically assumed the “living fossil” hypothesis (which I’ve told you is bunk), the results in 4 out of 5 cases concluded that the genome was “slowly evolving”; in 7 out of 7 cases in which the work was critical of the “living fossil” hypothesis or did not even acknowledge it, they found that coelacanth genes were evolving at a perfectly ordinary rate.
Research does not occur in a theoretical vacuum. Still, it’s disturbing that somehow authors with an ill-formed hypothetical framework were able to do their research without noting data that contradicted their ideas.
Maybe a start to correcting this particular instance of a problem is to throw out the bad ideas that are leading people astray. The authors strongly urge us to purge this garbage from our thinking.
Latimeria was first labelled as a ‘living fossil’ because the fossil genera were known before the extant species was discovered, and erroneous biological interpretations have grown and reports still show little morphological and molecular evolution. A closer look at the available molecular and morphological data has allowed us to show that most of the available studies do not show low substitution rates in the Latimeria genome, and furthermore, as pointed out by Forey [3] long before us, the supposed morphological stability of coelacanths from the Devonian until the present is not based on real data. As a consequence, the idea that the coelacanth is a biological ‘living fossil’ is a long held but false belief which should not bias the interpretation of molecular data in extant Latimeria populations. The same reasoning could be generalised to other extant species (such as hagfish, lamprey, shark, lungfish and tatuara, to cite few examples of vertebrates) that for various reasons are often presented as ‘ancient’, ‘primitive’, or ‘ancestral’ even if a lot of recent data has shown that they have many derived traits [58–64]. We hope that this review will contribute to dispelling the myth of the coelacanth as a ‘living fossil’ and help biologists keep in mind that actual fossils are dead.
But of course we also shouldn’t let that color our data. If analyses showed a significantly reduced substitution rate in the evolution of a species, it ought to get published. If nothing else, it would be an interesting problem for evolutionary theory. Coelacanths, though, don’t represent that problem.
Amemiya CT, Alföldi J, Lee AP, Fan S, Philippe H, Maccallum I, Braasch I, Manousaki T, Schneider I, Rohner N, Organ C, Chalopin D, Smith JJ, Robinson M, Dorrington RA, Gerdol M, Aken B, Biscotti MA, Barucca M, Baurain D, Berlin AM, Blatch GL, Buonocore F, Burmester T, Campbell MS, Canapa A, Cannon JP, Christoffels A, De Moro G, Edkins AL, Fan L, Fausto AM, Feiner N, Forconi M, Gamieldien J, Gnerre S, Gnirke A, Goldstone JV, Haerty W, Hahn ME, Hesse U, Hoffmann S, Johnson J, Karchner SI, Kuraku S, Lara M, Levin JZ, Litman GW, Mauceli E, Miyake T, Mueller MG, Nelson DR, Nitsche A, Olmo E, Ota T, Pallavicini A, Panji S, Picone B, Ponting CP, Prohaska SJ, Przybylski D, Saha NR, Ravi V, Ribeiro FJ, Sauka-Spengler T, Scapigliati G, Searle SM, Sharpe T, Simakov O, Stadler PF, Stegeman JJ, Sumiyama K, Tabbaa D, Tafer H, Turner-Maier J, van Heusden P, White S, Williams L, Yandell M, Brinkmann H, Volff JN, Tabin CJ, Shubin N, Schartl M, Jaffe DB, Postlethwait JH, Venkatesh B, Di Palma F, Lander ES, Meyer A, Lindblad-Toh K. (2013) The African coelacanth genome provides insights into tetrapod evolution. Nature 496(7445):311-316.
Casane D, Laurenti P (2013) Why coelacanths are not ‘living fossils’: A review of molecular and morphological data. Bioessays 35: 332–338.


I’m going to self-plagiarize from the other thread:
I’ll add that the argument here that modern coelacanths do so look different from the fossil ones–and also look how diverse the lineage used to be–are exactly as rigorous and sciency as the opposing view that they still look pretty much the same today. I look at the illustrated phylogeny and osteodiagram and I see a bunch of fish that all look a hell of a lot more similar to each other than any does to any other living fish (possibly excepting Allenypterus). I linked here on the other thread; it’s simply a judgment call.
As for the part that broke your brain, it’s sloppy writing and probably reflects misconceptions, but it’s not that tough to figure out what they mean. In an environment that is stable for very long periods of time, what’s missing are directional selection pressures. Morphological conservatism is attributed to the constancy of what evolutionary biologists call stabilizing selection pressures (= a constant environment). Nothing stops micro-evolution by drift and selection on new mutations, so small differences evolve, but no, you do not expect the accumulation of tons of neutral variation (well, you do if you’re Moran). It’s not brain-breaking, just poorly explained.
In my first year of Marine Biology my tutor set us the assignment to right a report and a presentation on an “evolutionary success story of the marine environment”. Not wanting to do what every other student had apparently done in the past ie sharks, dolphins and various plankton groups, I chose the coelacanth, reasoning that as a “living fossil” it must be getting something right. Also I like coelacanths and had a good excuse to learn more about them and then talk about them in an environment where my peers will be obliged to listen, which for someone like myself is not something to be sniffed at.
My research essentially produced this blog post and blew apart my previous ideas about coelacanths and the concept of living fossils. Which meant my initial justification for their being a “success story” no longer existed, but I’d already done all this research and I really wanted to write an essay on coelacanths. At this point I began to wonder what exactly “evolutionary success story” can possibly mean. Ultimately I reasoned that since some of them are still alive they must be pretty successful. I don’t know what other criteria could possibly matter.
“slow rate of coelacanth evolution could be due to a lack of natural-selection pressure” is hard to resolve with “stabilizing selection pressures”. You’re arguing for a lot of selection to retain a particular morphology; Lindblad-Toh is talking about a lack of selection. That’s remarkably bad writing to say the opposite of what you think she means!
But are coelecanths creationists, or not as deluded as Ken Ham?
Glen Davidson
As a very much non-scientist, I have two things to say:
1) This was a very cogent explanation of the errors in a widely-held belief (I had previously bought the “living fossil” argument)
2) I really, really want an Allenypterus plushie.
If evolution is true, why are there still coelacanths?
If you’ve ever been to Maui, and specifically up the Haleakala volcano, you’ve seen silverswords, those weird silvery yucca-looking things that look like they belong on a desert planet in one of the old Star Treks. Silverswords are at the opposite end of the spectrum from coelacanths, i.e. where coelacanths have “evolved” just fine (i.e. accumulated mutations relative to the ancestor) but happen to still look a lot like those ancestors, silverswords look completely different from California tarweed but only separated 5-10 MA ago and aren’t that genetically dissimilar (you can back cross them). And they look nothing alike. When we just had fossils and morphology to go on we were a lot more limited.
as explained there are no “living fossils” all things are always evolving and adapting to the various selecting pressures they experience.
The question that comes to mind is how or why do some groups completely disappear from the living present and some like the one discussed here have only one survivor.
uncle frogy
PZ,
The authors do note that only the protein coding genes have reduced substitution rates, not the whole genome:
“In this study, we have confirmed that the protein-coding genes
of L. chalumnae show a decreased substitution rate compared to those
of other sequenced vertebrates, even though its genome as a whole
does not show evidence of low genome plasticity.”
Also, they state the following:
“The reason for this
lower substitution rate is still unknown, although a static habitat and a
lack of predation over evolutionary timescales could be contributing
factors to a lower need for adaptation. A closer examination of gene
families that show either unusually high or low levels of directional
selection indicative of adaptation in the coelacanth may provide
information on which selective pressures acted, and which pressures
did not act, to shape this evolutionary relict.”
Allenypterus and Mawsonia are adorable.
The claim that the coelacanth is solowly evolving because selection pressure in the deep ocean is not as great sials close to a classic creationist trope. That is noe of the organism that went extinct , (in the oceans at least) are really extinct. They are just lurking away in the ocean deeps. Louis Agassiz subscribed to this view and mounted several deep sea trawling expeditions in a fruitless attempt to prove it.
Another creationist twist on this is that everything was created at once and the progressive extinction and replacement of organisms seen in the fossil record was only due to replacement by already organisms recolonizing shallow seas from their deep ocean hiding places. This idea is proposed in “the Transformist Illusion” by Douglas Dewar.
Hmm Pterosaurs and T.Rex living it up on the bottom of the Atlantic waiting for there turn to make landfall. “Atlantis meets Jurassic Park”, now that would make a good B grade sequel
@Sassafras, #10:
So are Hadronector and Macropoma. They ought to have little word balloons with smiley faces in them.
So, given the small sample size and all that, this isn’t evidence of that much, but as an allegory for why science (messily) works, I love it. 11 out of 12 groups found evidence to match their preconceptions, but 1 out of 12 found and published evidence that contradicted it (and it was one of the groups whose preconceptions were misguided). Just like any other pursuit, science tends most of the time to be people reinforcing their own beliefs — but there’s just enough pull towards reality that over time it tends to sift out the truth. Love it.
When it comes to morphological conservatism, I think you’re missing that this is a comparative issue. Coelacanths are extremely morphologically conservative compared to their sister clade. That doesn’t mean they don’t have any morphological diversity, of course–they do, as you point out. However, compared to the clade of lungfishes + tetrapods, there’s just not much going on in coelacanths.
I’m not sure this is really all that interesting or exciting a comparison, but when people talk about morphological conservatism in coelacanths, I’m pretty sure they don’t mean “all coelacanths are exactly the same” but rather that tetrapods have diversified spectacularly and coelacanths haven’t.
no it’s not. It’s obviously what they meant: a lack of directional selection pressures (of the type that drive change and adaptation to changing environments).
People just don’t realize (and I mentioned ‘misconceptions’) that mostly what selection does is to
the current level of population-wide adaptation.
well, that’s a blockquote for emphasis there.
always preview.
A minor quibble, that actually makes your point stronger, is that there are in fact two living species of coelacanth both in the same genus. The one found off the east coast of Africa Latimeria chalumnae and the one found off Indonesia L. menadoensis.
I also remember reading a paper at some point that found genes for morphology showed little differentiation between populations, but other gene regions accumulated mutations well within the range of vertebrates. Which is consistent with the stabilising selection hypothesis. Unfortunately, I can’t find that paper now…
Whenever I see the word coelacanth all I can think about is Dirk Gently and the Rhyme of the Ancient Mariner…
I award Leela Moses a shiny new Internetz
— — — — — — —
What about non-vertebrate chordates? They have not changed very much, so apparently they are a success story, compared to those vulgar vertebrats who respect no traditions.
(vertebrats was a spelling error, but it works in the context)
“(vertebrats was a spelling error, but it works in the context)”
I just realised I have created a successful mutation! Now, on to re-creating the works of Shakespeare.
Allowing as how we know gamma radiation can alter DNA, somewhat randomly, is there any evidence that creatures who live shielded from the effects of cosmic radiation deep undersea undergo less mutation/change, relative to us surface-dwellers? If I were to guess, cosmic radiation might be a very small factor. (PS I am not a biologist, if you hadn’t guessed!)
Holy crap. The fin of Shoshonia looks a lot like the preaxial side of a lungfish fin.
It is. A bigger factor is whether you live on granite (which contains plenty of radioactive stuff) or, say, clay. Besides, of all known coelacanths, only Latimeria lives in the deep sea; all others lived in shallow waters or even freshwater.
The biggest source of mutations is the imperfection of the DNA-copying and the DNA-repairing machinery.
Jerry Coyne:
PZ Myers:
Which informed opinion is wrong?
Neither. The rate of evolution is what selection makes out of the rate of substitution. Substitution (and other kinds of mutation) create diversity, and then selection cuts it down – in different ways for different environments.
If anyone is interested here are two links.
A brief summary of why the authors think this paper is important.
http://phenomena.nationalgeographic.com/2013/04/22/a-most-amazing-fish-join-our-google-hangout-about-coelacanths-on-thursday/
And an invitation to join a google hang out to discuss the paper and the scientific community’s response, including PZ’s.
https://plus.google.com/u/0/events/cf6oq1bjafkuag6h23l2vmevmuk
Maybe I’m missing something but Coyne is agreeing with the authors of the recent paper that Coelacanth genes (the ones looked at in the recent study) show reduced rate of evolution.
Again, PZ is not talking about rates of evolution, but rates of substitution.
My confusion came from this sentence, which I thought was implying: therefore gene evolution rates.
Nevertheless, Coyne agrees generally with the paper’s conclusions (including reduced substitution rates) and Myers does not. I thought I had pinpointed the source of disagreement.
That’s the usual sloppy usage of “substitution rate” for “what’s left of the substitution rate once selection is done with it”.
As PZ has pointed out, this doesn’t make sense: weaker natural selection should leave more, not less, of the substitution rate visible.
“A static habitat”, on the other hand, could exert strong stabilizing selection; but it’s only been static since at most the split between Latimeria and Macropoma.
IIRC most “ancestral” coelacanths likely lived in shallow waters, many in freshwater at that.
Also, relative to the oceans (and to most marine biologists) coelacanths don’t live that “deep” – the South African population generally hangs out at around 130m during the day (feeding in shallower water) with the odd report of coelacanth sightings at around 50m, whilst those in the Comoros Islands rest in caves at around 300m, and I recall L. menadoensis from about 150m. The mean depth of the ocean is approximately 2,000m, and “deep” only really starts at 700-1,000m. Little is known from the other African countries where it has been caught.
That might be pretty “far down” for naked (or even suit-clad) humans, but it isn’t for marine life.
The later pages of Forey’s History of the Coelacanth Fishes (notably from about 367 onward – ch 12) support the extant Latimeria being relatively “advanced” and not at all a “fish out of time” – it is a fish of its time! Effectively, this lends weight to the discarding of the “living fossil” tag, which will cause scientific communicators to scramble for another bite-sized label to fit this fish, particularly in light of the 75th anniversary of its scientific discovery this year.
I guess this also perhaps lends yet more weight to the view of Nature being increasingly a popular science magazine rather than a serious academic journal!
Oh no, Nature isn’t popular. It’s an extended-abstract publication, like Science, which publishes slightly longer abstracts, and PNAS, which allows yet a bit more. The recent giant mammal phylogeny in Science had “supplementary information” (actually the real paper) which had over 100 pages and even its own supplementary information!