All life on Earth is carbon-based which of course makes this molecule of considerable interest for scientists. A carbon atom has 6 electrons which means that it has four electrons in the valence shell and results in it being able to form different kinds of chains and combinations, such as buckminsterfullerenes (often shortened to just ‘fullerenes’, more popularly known as ‘buckyballs’) that consists of 60 carbon atoms in the shape of a geodesic sphere.
Now comes along a report that scientists have created a ring that consists of 18 carbon atoms. Other carbon molecules such as fullerenes, graphene, and carbon nanotubes have each carbon atom linking to three other carbon atoms. In this new molecule, each carbon links to just two other carbon atoms, enabling them to form rings called cyclocarbons.
Theory had suggested that C18 is the smallest ring of carbon that is stable but creating it from the ground up had proved to be difficult. So a group of scientists tried a different tack, of going from the top down. They started with the molecule C24O6 and then proceeded to successively knock out pairs of CO (carbon monoxide) molecules, producing C22O4, C20O2, and finally C18.
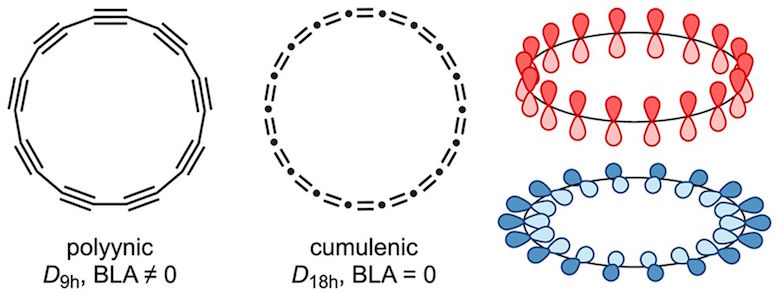
The big unanswered question had been whether these rings would have what is known as a polyynic structure with alternating triple and single bonds or would be cumulenic, with pairs of double bonds.
Now that the researchers had an actual molecule to study, they could answer the question.
And having finally created a stable version of the thing they could study, the researchers put to rest a long-standing debate about what kind of atomic bonds the carbon atoms within such a molecule would share. The ring is made up of alternating triple and single bonds, known as a polyynic structure, the researchers found. Amazingly, they confirmed this by studying how the molecules looked. They literally compared photos of the individual collections of atoms to expected theoretical models.
You can read the paper in the journal Science here.

Manu,
quick check -- surely a total of 6 electrons per atom? 4 in the valance shells (p) is still the relevant bonding bit.
Ross
The question is, would C18 form in nature?
After C60 was discovered, scientists realized it occured in stars and arc lamps (burning carbon tips). I can’t find an image of it, but there’s a graph that shows the frequency of carbon structures larger than 8. C60 and C70 were the most common, but pairs of carbon atom scan be added or removed, going as low as C48 before collapsing. The creation of carbon nanotubes shows there is no upper end to how many carbon atoms can be uaed.
A NOVA episode on Buckyballs, from 1995 is on youtube:
Ross @#1,
You are of course correct. Thanks for pointing out my error that I have rectified.
…known as a polyynic structure…
Which I read at first as polycynic, a
usefulnecessary formulation for our interesting times.This is cool! I wonder, how wave-y this ring is. And how strained it is.