While we’ve been waiting and waiting for the physicists to get their act together and deliver on Mr Fusion home energy sources and flying cars, the biologists have been making great progress on the kinds of things that turn biologists on. The latest development: growing tiny little human brains in a bucket. Only let’s not call them brains…they are cerebral organoids. Hugo Gernsback would be so proud.
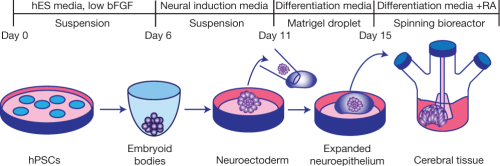
Here’s the latest development. Start with embryonic human stem cells, or induced pluripotent stem cells (cells which you’ve reset to a kind of embryonic state by using a virus to transfect them with the genes OCT4, SOX2, KLF4 and MYC, which trigger a change to a pluripotent state). Culture them in a cocktail of chemicals like basic fibroblastic growth factor and retinoic acid, which induces the cells to become neurectoderm, a precursor tissue of the nervous system. Imbed these cells in a gelatinous capsule that gives them a framework on which to grow, and also prevents them from just sprawling out into an amorphous neurectodermal sheet. Let them grow in a spinning bioreactor which circulates nutrients around them, and watch. They begin to form structures resembling those of the embryonic human brain, all by themselves.
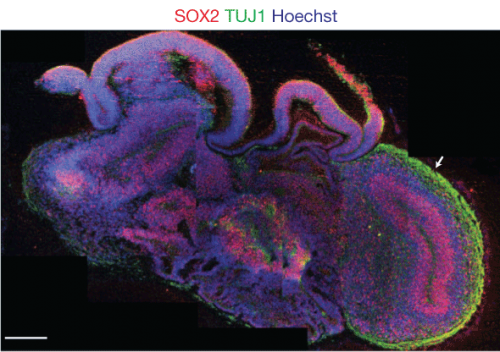
They show many of the properties of normal embryonic brains. Brains develop from the inside out; new neurons arise deep inside, and then migrate outwards along radial glia to the surface. These mini-brains show similar behavior, forming the beginnings of a laminar structure with roughly the same pattern of growth. They exhibit regional specification. We have a forebrain, midbrain, and hindbrain, for instance, and there are molecular markers for these areas; those molecular markers are selectively expressed in areas of the mini-brains, too. During development, our neurons exhibit fleeting electrical activity, especially the production of calcium action potentials (we gradually switch to sodium action potentials as the nervous system matures). These brains have bursts of calcium activity which can be diminished with tetrodotoxin, a nerve poison that affects signal transmission in neurons.
Awesome. Before you start imagining growing complete adult human brains in a vat to the point where they start doing philosophy, though, there are realistic limitations.
While there are regions expressing markers for typical human brain regions, they aren’t well organized — I looked at the sections of cerebral organoids, and while bits and pieces looked familiar, they weren’t in their canonical relationships to one another. It’s a kind of scrambled brain.
The laminar structure of the brain doesn’t fully form — it’s just the rough beginnings. It really is like a very early embryonic brain, and is not going to function to generate thoughts and perceptions.
It’s only brain tissue. They aren’t growing elements of the circulatory system, for instance, so there are no blood vessels delivering nutrients. That limits growth, and the largest cerebral organoids are only about 4mm in diameter. That might only be enough to generate an assistant professor of philosophy. (I joke! Don’t come after me, philosophy fans.)
Now this is pretty darned cool, and would be a shoo-in to win first prize at the Mad Science Fair, but you might be wondering what you can use it for. These are not functional brains, so no, you militarists, you can’t use them to control cruise missiles. What they are good for is studying developmental processes that build human brains (pure science!) and for figuring out the mechanistic causes of serious brain disorders (medical science!).
And the authors turned around and started doing just that. There are known defects that affect the proliferation of cells building structures in the brain, genetic diseases like microcephaly. You cannot do experiments on microcephalic human beings, and it’s been very difficult to generate good animal models of microcephaly — we have such unusual brains to begin with that it’s hard to find a brain analogous in sufficient detail in mice. But here’s what they can do.
They had a patient with microcephaly, with a known genetic cause (a mutation in a gene called CDK5RAP2). They can’t experiment on his brain, obviously, but what they could do is take a few of his skin cells, transfect them with the four inducing genes, and produce a clone which could be cultured in a dish and put through the organoid production procedure and make little tiny copies of his embryonic brain state. Now you can do experiments.
What they observed was that some brain areas in the organoids were smaller (complicated by the fact that overall growth was reduced), and that there more more neurons and fewer glia in affected regions. The hypothesis is that CDK5RAP2 maintains cells in a dividing state, and it’s absence causes premature maturation of neurons, which leads to reduced total numbers of neurons in the adult. They also tried inducing greater CDK5RAP2 in the organoids to rescue the phenotype — the experiment was confused by the fact that CDK5RAP2 overproduction seems to trigger cell death, and they do not have precise control over dosages, but they do find suggestions that glial production is rescued. They also did the complementary experiment of RNAi knockouts of CDK5RAP2 in non-patient organoids, and they did see a surge of neuron production. So it looks like they’re getting a good handle on the cellular processes behind this form of microcephaly.
So what they’ve built is a useful model for studying early brain development in humans that doesn’t involve experimenting on any actual humans. This is going to be useful for all sorts of developmental disorders.
Also, Mad Science Fair contests.
Lancaster MA, Renner M, Martin C-A, Wenzel D, Bicknell LS, Hurles ME, Homfray T, Penninger JM, Jackson AP, Knoblich JA (2013) Cerebral organoids model human brain development and microcephaly. Nature doi:10.1038/nature12517.


Waiting for the anti-choicers to freak out about this…
Estelle Reiner: “I’ll have what she’s thinking.”
Awesome.
Typo alert:
/copy editor
Couldn’t they build a network of porous tubing first, in the nutrient tanks, and then grow the brain tissues around it?
Happens to all of us.
This is your brain.
This is your brain on drugs.
This is a brain grown in a vat using the same sort of hydroponic technology used to grow drugs.
Any questions?
Well, at least we have a low-impact means of feeding the zombies when they start wandering around looking for Braaaaaaaainnnnss…. in October.
Mmmm…. scrambled brains. tastes like creationist.
Wonderful. And yes, I’m amazed that the religious anti-stem-cell-research brigade aren’t joined by the anti-choicers in paroxysms of apoplexy.
…so this means that they’ll have brand new brains for us to download ourselves into when….
No?
[This is pretty damn cool anyways :D]
PZ @ 1;
Don’t forget the more generic godbotherers and anti-intellectuals who will almost certainly trot out that old scaremongering stand-by about the dangers of scientists ‘meddling with things humanity was never meant to know!’ (Cue ominous thunder and lightning in the background)
Then there will be your paranoid conspiracy theory types who will say something like ‘if the government is prepared to admit that they can do this, what is the top secret stuff like, huh? Area Fifty One = Frankenstein Central people! Run for your lives!’
They’ll almost certainly ignore/not know the fact that Dr Frankenstein was the name of the scientist in the story, not the monster. They will probably also skip over all the stuff about alienation and the monstrousness of ordinary people that is a central theme of the tale – that wouldn’t make for good delusional ranting, afterall.
On the subject of Frankenstein’s monster, there will of course be the anti-GM protestors who will say ‘we told you so; first it was Frankenstein Foods, and nobody listened to our warnings. Now look what teh ebil scientists are doing!’.
I expect we will see a fair bit of figurative gnashing teeth and rending of clothes about this. We will probably see another round of attempts to ban this type of research and restrict the intellectual freedoms of scientists as well, since there are always clueless twits who assume there are no such things as reseach ethics boards and laws regulating biotechnology, and that what the debate really needs is less informed input from actual specialists in the field who have spent decades accumulating hard won data to back up their arguments, and more screeching from people who assume that all biological research is done by evil super-corporatrions that look like a cross between Weyland Yutani and Umbrella Corp.
Darn – I missed the anti-stem cell research lot, but opposablethumbs covered them @ 8.
Stop using your brain for thinking my thoughts.
I, for one, welcome our new cerebral organoid overlords.
am I understanding this correctly ? the brain develops with the blood supply and starts out like this unorganized tissue and over time increases its organization as it reacts to more inputs from the rest of the nervous system.
that there is a relationship between its organizational complexity and the activity of the brain that works both ways. the more activity the more complexity and the more complexity the more complex the activity
.
uncle frogy
I was thinking exactly what Dalillama said @4. Now combine that with unclefrogy. Hook it up to a computer to stimulate it and we may end up with Asimov’s positronic brain. Where is Dr. Susan Calvin?
Bodies! Bodies with no brains (and no, NOT Republicans) is what they need to be working on. I’ve just about hit my shelf-life – if you believe the statistics I probably have 20 years left, max – and I need an empty shell to slap my fully-formed me-brain into.
Seriously, this is some really cool stuff, but I don’t understand this: “These brains have bursts of calcium activity which can be diminished with tetrodotoxin, a nerve poison that affects signal transmission in neurons.” My first thought would be that you would want neuron transmission. Perhaps I should go try and find the article.
What they’re describing is an experimental test to determine if the observed calcium activity is truly related to neuronal transmission-related activity (in another source on this I recall reading that they do not think the neurons in these brainlets have yet made significant networks amongst themselves).
So, if (as hypothesized) the observed activity is due to the neurons doing something with their calcium channels, then exposure to tetrodotoxin will block it. If not (as per null hypothesis), then tetrodotoxin would not affect the observed calcium activity, and it would suggest that the observed activity was not related to neuron activity.
Since we observed a positive (ie, blockage of calcium activity by tetrodotoxin) we can infer this as evidence that the observed evidence really is due to the activity of calcium channels in the neurons, in keeping with the kinds of activities that calcium channels get up to in normal brains.
In other words, they are demonstrating that these experimental constructs demonstrate at least some of the normal activities of normal brains.
From here: http://phenomena.nationalgeographic.com/2013/08/28/the-cerebral-organoid-a-lab-grown-model-brain/
So basically, no actual significant amounts of information processing by the neurons, or, colloquially, nothing that yet approaches “thoughts”.
At least not yet….
Amphiox – Thank you so much! That answers my question completely and makes perfect sense.
Unclefrogy – What this paper is showing you is the self-organizing complexity of the brain that arises before there is much connectedness and activity of the neurons.
The brain and spinal cord start out as a flat sheet of dividing cells. These cells all look pretty much alike, but there is a body plan that distinguishes head from tail, top from bottom, and so on. This map causes different rates of division in various areas, followed by different neurons being born in those areas in a set order. This paper shows a lot of this happening in self-organizing brain cells; the basic events don’t require the rest of the body. (Pretty amazing to me!) The calcium assay shows that eventually, individual neurons become active but there is no widespread connectivity yet.
You are correct that later on, neuronal connections and activity induce greater complexity of structures and networks. But the early organization does not require that.
I hope this helps; brain development is too complicated for me to describe simply.
Will they be a proper attractant for a Giant Zombie Zapper?
Look, they’re way ahead of you. Just give it some time. They’ve predicted brains (scrambled and non-scrambled, along with anything else you can think of) could occasionally be assembled just due to statistical fluctuations, which sounds like a lot less work than what you’re describing. Sure, some predict it won’t ever happen because it takes too much time, but they want the universe to be destroyed. (Totally not a quote-mine from Sean Carroll himself: “we need to destroy the universe.”) And those are apparently your only options. So whose side are you on?
… Anyway, this looks like some amazing research. I’m curious about how much or what kinds of “regional specification” there is here, but I’m more ignorant than I am curious. You mention forebrain, midbrain and hindbrain, but to me it just looks like a mess, so I guess I don’t understand what kind of significance the regions identified might have.
If you squint a little bit, you might envision that it is trying to develop into a superior form of life. I definitely see the beginnings of a mantle and tentacles.
Allow me:
Lawrenceville Plasma Physics Accelerating Advanced Fusion Energy
Not entirely ready to go, either, but looks kind of promising to this particular amateur charlatan at least. 5 MW non-radioactive, aneutronic cyclical focused fusion generator the size of a standard freight container for maybe $300k. Seems they are at the proven proof of concept stage now, establishing pulsed fusion bursts feeding He-ion beams into generator at near net energy profit.
.
Also not exactly flying cars, but have you checked out Tesla Model S with the transnational, free super charger network build-up? (Oops, that was for Norway.) Everybody already has an electric outlet in their garage, right.
.
And Elon the electric car guy’s raw sketch of a Hyperloop? $20, 35 minutes ride LA to SF. (His reusable rocket is probably off topic here.) https://twitter.com/elonmuskhttps://twitter.com/elonmusk/status/368461203317612544
Baby steps. Must be taken first.
…and it’s absence causes premature maturation of neurons…
PZ, do you think we could rewire human brains to prevent apostrophe misuse?
And it remains uninformed by any sensory input from the world with which to cogitate over…even assistant philosophy professors have that. 8)
OK I want to know how much the first synthetic brainburger will cost to make.
Also, forget flying cars. Its always been a silly pseudo-tech fantasy, hardly worthy of decent science fiction or futurist speculation. As long as human populations are concentrated in cities, any commuter system based on them is an inherently dangerous idea. We already have enough trouble controlling air traffic consisting of flying buses called ‘airplanes’.
But airplanes are fl–
And we already have a bunch of them which aren’t the size of commercial jumbo jets. I’d say the smaller ones are less like a flying bus, more like a flying car or flying motorcycle. (But we’ll probably never have flying trains … I guess because the tracks would be nonexistent too quickly?)
I think some people mean they just want them to cost as much as a car or maybe that they want more cupholders.
addresses newly created cerebrate
Awaken my child, and embrace the glory that is your birthright. Know that I am the Overmind, the eternal will of the Swarm, and that you have been created to serve me.
(If those words haven’t been uttered in the lab I’ll eat my saves.)
cr: Yes yes, they are obviously already with us. They’re called private planes. And planes as inexpensive as a car already exist too: ultralights. But I for one would not look forward to millions of commuters piloting and navigating their craft under the direction of air traffic controllers. It would interfere with their texting.
Billions, not millions. Lots of texting to do.
But we wouldn’t need them because we’ll have robots to do it all for us making the necessary sprockets and cogs and so forth (but none of the fun jobs like working at a factory mindlessly pushing buttons). And we’ll live in the sky anyway, so landing isn’t really such a big deal. Don’t you know about the Jetsons? (When they met the Flintstones, yes, that was a documentary. I don’t know why people keep asking me.)
@naturalcynic #23:
“Only one cell in a billion was fit to be nurtured.”